In recent years it has become clear that redox changes are also important regulators of cellular function, with several redox species, for example H2O2, serving as key second messengers in cellular signaling pathways. It is now apparent that there is no such thing as a ‘cellular redox state’. Instead, changes in specific redox species, in specific subcellular locations at specific time points mediate specific cellular responses. However, our knowledge of cellular redox dynamics remains extremely limited. The majority of redox research to date has relied heavily on small chemical probes, which have poorly defined redox species specificity. Furthermore, they offer very limited spatial and temporal resolution, it is almost impossible to determine in which subcellular location the redox change occurred nor when or how long‐lasting the change was. A major advance in our ability to study cellular redox processes came with the development of genetically encoded redox probes. These probes allow the real‐time measurement of defined redox species, with subcellular compartment resolution, in intact, living cells. Of particular value are the Grx1‐roGFP2 (for measuring the glutathione redox potential (EGSH); arguably the most important small molecule thiol compound in the cell) and roGFP2‐Orp1 (for measuring changes in H2O2 levels; arguably the most important reactive oxygen species in the cell) probes.
In our lab we make use of the redox sensors and the newly developed transgenic Orp1-roGFP2 mice, to study the role of cellular reduction-oxidation (redox) changes in pancreatic islets during the progression of insulin resistance to diabetes. In particular, we aim to understand how different signaling molecules (ROS, Ca2+, NAD(P)H) influence insulin secretion and beta cell (dys)function in health and disease. More recently, as a part of an approved funding between Aachen and Homburg (SFB TRR219), we have also started to study the causal role of redox changes in cardiac myocytes remodeling, secondary to Chronic Kidney Disease (CKD).
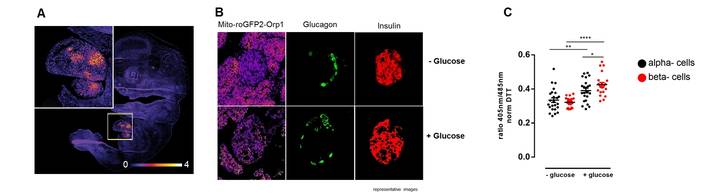 |
| Redox histology. (A) Mouse embryo E14.0 show increased mitochondrial sensor oxidation in the liver. (B) Islets from mt-roGFP-ORP1 mice shows more oxidized alpha and beta cells after glucose challenge. |




